Breast cancer is the most frequently diagnosed life-threatening cancer in women and the leading cause of cancer death among them worldwide. It includes a heterogeneous collection of diseases with various histologically defined subsets, clinical presentations, responses to treatment and outcomes. We describe a case of female patient with ductal breast carcinoma.
Are the case reports useful for you? Would you also require case reports from other clinical disciplines? Do you have any comments or suggestions for improvement? Please provide your feedback by filling out a short questionnaire.
Anamnesis:
57-year-old patient presented at the surgery department of our hospital and reported that 2 years ago she had felt a small lump in her right breast; over the last 4 months the lump had become bigger and she noted right axillary tumour, too. She had no other medical problems. She had no personal or family history of breast or ovarian cancer. Her anamnesis included G3P3, menarche, at 11 years, and menopause at 46. All 3 children were all healthy. No other important anamnestic data were reported. On physical examination a 2-cm mass at 2 o'clock on her right breast were observed and axillary pathological node involvement.
Imaging methods:
A diagnostic mammogram and ultrasound image showed a spiculated density corresponding to the palpable mass and a hypoechoic mass measuring 2,3 x 1,9 x 1,7 cm; no abnormalities were seen in the left breast. A core biopsy of the breast was performed immediately and showed high-grade infiltrating ductal carcinoma, estrogen receptor (ER)-positive, progesterone receptor (PR)-negative, and HER2-negative. She was sent to lymphoscintigraphy (Figure 1, 2) - Sentinel lymph node mapping. It is an imaging technique used to identify the lymph drainage basin, determine the number of sentinel nodes, differentiate sentinel nodes from subsequent nodes, locate the sentinel node in an unexprected location, and mark the sentinel node over the skin for biopsy (Figure 3).
Therapy:
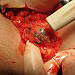
Patient received a combination of radiocolloid injection one day before surgery and blue – dye injection into the mark (which was drawn on the skin above the tumor during lymphoscintigraphy) immediately before surgery (Figure 4, 5). It gives the highest sensitivity and specificity of the technique. Round incision around nipple was done, and tumor with safe borders was excised and sent to pathology (Figure 6-9). Gamma detecting probe was covered with a sterile cover for use within the surgical field. Sentinel node was detected by high frequency of sounds coming out from the machine and high number of radioactivity on display (Figure 10-12). Sentinel node was visible, too - it is blue-infiltrated node with dye injection (Figure 13).
Sentinel node biopsy was sent during surgery to the pathology, that revealed a cancerous node (Figure 14). Surgeon decided for exenteration (Figure 15). Drain was placed in the field after resection of the tumour and in the space after exenteration of lymph nodes. Wounds were closed with continuous stitches (Figure 16). Two of 13 nodes were cancerous according to definitive histology. Borders around tumour mass were clear from cancer cells. The patient was transferred to the oncology department, and after being presented to the breast committee, was started on analgesics, chemotherapy, radiotherapy and endocrine therapy. She was treated with dose-dense doxorubicin and cyclophosphamide followed by local radiotherapy and adjuvant oral endocrine therapy. Now, approximately 3 years after surgery, all other parameters including renal and liver function were normal. She is further evaluated with CT of the chest, abdomen, and pelvis with and without contrast revealed no other secondary deposits. The patient has no symptoms and has returned to her previous employment full time.
Discussion:
Breast cancer is a heterogeneous disease - 100 different types of malign tumors. The most frequent malignant disease in the case of women. Risk factors: age (50-65 years), hormonal status : -early menarche, late menopause, long HAK , late primigravidity, low amount of gravidity, positive family anamnesis: BRCA gens, frequent X ray and CT of chest and obesity, stress, depressions.
Treatment should be provided by a multidisciplinary team, which includes: at least one surgeon, radiation oncologist, medical oncologist, radiologist and pathologist, and plastic surgeons, who are specialised in breast cancer. Screening: Mammography screening, every 2 years, in the age group of 50–69 years. There is controversy and no consensus regarding the role of screening in women aged 40–49 years, or for the use of ultrasound. In women with familial breast cancer, with or without proven BRCA mutations annual screening with magnetic resonance imaging (MRI) of the breast, in combination with mammography, can detect the disease. I tis recommend annual MRI concomitantly or alternating every 6 months with mammography, starting 10 years younger than the youngest case in the family.
The diagnosis of breast cancer is based on clinical examination in combination with imaging, and confirmed by pathological assessment. Clinical examination includes bimanual palpation of the breasts and locoregional lymph nodes. Other assessments include: complete personal medical history, family history relating to breast/ovarian and other cancers, a full blood count, liver and renal function tests, alkaline phosphatase and calcium levels. Imaging includes bilateral mammography and ultrasound of the breast and regional lymph nodes. An MRI should be considered in cases of: familial breast cancer associated with BRCA mutations, breast implants, lobular cancers, suspicion of multifocality/ multicentricity (particularly in lobular breast cancer), or large discrepancies between conventional imaging and clinical examination. CT scan of the chest, an abdominal ultrasound or CT scan and a bone scan can be considered for patients with: clinically positive axillary nodes, large tumours (e.g. ≥5 cm), aggressive biology or clinical signs, symptoms or laboratory values suggesting the presence of metastases. Several new techniques are being tested for screening and diagnostic imaging, such as: 3D mammography (breast tomosynthesis), 3D ultrasound, shear wave elastography and contrast-enhanced mammography/ spectral mammography. Pathological diagnosis should be based on a core needle biopsy, obtained preferably by ultrasound or stereotactic guidance.
A core needle biopsy (if this is not possible, at least a fine nedele aspiration indicating carcinoma) must be obtained before any type of treatment is initiated. A marker (e.g. surgical clip, carbon) should be placed into the tumour at biopsy, to ensure surgical resection of the correct site. Final pathological diagnosis should be made according to classification and the tumour–node–metastases (TNM) staging system. The pathological report should include the histological type, grade, immunohistochemical evaluation of oestrogen receptor (ER) status, evaluation of progesterone receptor (PgR) and human epidermal growth factor 2 receptor (HER2) gene expression, the number, location and maximum diameter of the tumours removed, the total number of removed and positive lymph number of involved regional lymph nodes. Arguably the major change in the surgical treatment of primary breast cancer has been a shift towards breast conservation treatment - wide local excisio, radiation and chemotherapy. In some patients, mastectomy is still carried out due to: tumour size (relative to breast size), tumour multicentricity, inability to achieve negative surgical margins after multiple resections, prior radiation to the chest wall/breast or other contraindications to RT, or patient choice. For patients undergoing wide local excision, no tumour at the inked margin is required and >2 mm (for in situ disease) is preferred. Marking the tumour bed with clips in a standardised way facilitates accurate planning of the radiation boost field, if it is indicated.
For women undergoing breast reconstruction, whether immediate or delayed, a wide range of surgical options are available. Silicone gel implants are safe and acceptable components of the reconstructive armamentarium. Advances in gel cross-linking have reduced silicone bleed, and cohesive gel implants are likely to have fewer problems relating to capsular rupture. Autologous tissue flaps can replace relatively large volumes of breast tissue. Tissue can be taken from the latissimus dorsi muscle from the back, transverse rectus abdominis muscle, the free deep inferior epigastric perforator flap from the lower abdomen, superior gluteal artery-based perforator flap or free gracilis-based flap.
Regional lymph node status remains one of the strongest predictors of long-term prognosis in primary breast cancer. Axillary clearance is associated with lymphoedema affecting the upper limb in up to 25% of women following surgery. In patients with clinically and imaging negative axilla, the best timing to carry out sentinel lymph node biopsy (SLNB). SLNB, rather than full nodal clearance, is now accepted as the standard of care for axillary staging in early, clinically node-negative breast cancer [II, A], unless axillary node involvement is proven on ultrasound-guided biopsy. With appropriate training in the dual radiocolloid/blue dye, high identification rates (over 97%), low false-negative rates and favourable axillary recurrence rates following SLNB are achievable. SLNB delivers less morbidity in terms of shoulder stiffness and arm swelling. Patients with involvement of the sentinel lymph node, undergo exenteration of lyphatic nodes from axilla, irradiation and adjuvant systemic treatment. Risk-reducing mastectomy: Risk-reducing surgery (with prophylactic bilateral mastectomy and reconstruction) may be offered to women at very high risk, such as those who have had previous chest irradiation for lymphoma or BRCA1 or BRCA2 gene mutation carriers. The decision on systemic adjuvant treatment should be based on the predicted sensitivity to particular treatment types, the benefit from their use and an individual’s risk of relapse. The final decision should also incorporate the predicted treatment sequelae, the patient’s biological age, general health status, comorbidities and preferences. Treatment should start preferably within 2–6 weeks after surgery. Endocrine therapy is indicated in all patients with detectable ER/ PgR and HER2 expression (defined as ≥1% of invasive cancer cells).
Authors declare the case report will not be published in any national or international publications.
Classification ICD-10:
D05 Carcinoma in situ of breast
4-D assessment:
Textbooks and manuals |
Educational websites and atlases |
Digital video |
Presentations and animations |
Casuistics in images |
E-learning courses (LMS) |
Unreviewed |
Undergraduate level |
Graduate |
Advanced Graduated |
Complex |
HodnoceníPlease select achieved education degree and then evaluate the teaching material particularly in light of material suitability for self-learning.
Student – student of bachelor or master degree
Graduate – graduate of bachelor or master degree PhD. Graduate – Ph.D. student, Ph.D. graduate, researcher, ...
%
Evaluate evaluatethis contribution first! |
 Contribution content is subject to licence Creative Commons Uveďte autora-Neužívejte dílo komerčně-Nezasahujte do díla Attribution 3.0 Czech Republic
Contribution content is subject to licence Creative Commons Uveďte autora-Neužívejte dílo komerčně-Nezasahujte do díla Attribution 3.0 Czech Republic
citation: Martina Vidová Uğurbaş, Lucia Sukovská Lakyová: Management of breast cancer. Multimedia support in the education of clinical and health care disciplines :: Portal of Pavol Jozef Šafárik University in Košice Faculty of Medicine [online] , [cit. 15. 03. 2026]. Available from WWW: https://portal.lf.upjs.sk/articles.php?aid=290. ISSN 1337-7000.